We’re conscious many in the commercial vehicle and road freight sector are having to get to grips with a whole new world of technology, terms and vehicle operating conditions. In the first of an occasional series of explainer articles, Andy Salter takes a look at how Lithium-ion batteries being used to power battery electric HGVs work
Having spent the last 30 years writing about diesel-powered trucks, shifting to carbon zero heavy duty trucks has meant a steep learning curve. Over the coming weeks and months I’ll be writing a series of explainer articles picking up on some of the key components used in a battery electric or hydrogen fuel cell lorry. Don’t worry, I only scraped a pass in O-level Physics, these articles won’t be too in-depth!
At the heart of every Battery electric truck is the battery, think of it as the fuel tank - its the energy storage and transport medium. Credit for inventing the battery lies with Alessandro Volta (see the link to the truck company, clever eh?) back in the late 1700s and over the ensuing years the battery has increased its importance. From lead acid batteries in cars, through Nickel rechargeable batteries in power tools and alkaline batteries used pretty much everywhere around the home, the automotive sector has now settled on the Lithium-ion battery as the cell of choice.
A battery is made up of an anode, cathode, separator, electrolyte, and two current collectors (positive and negative). The anode and cathode store the lithium. The electrolyte carries positively charged lithium ions from the anode to the cathode and vice versa through the separator. The movement of the lithium ions creates free electrons in the anode which creates a charge at the positive current collector. The electrical current then flows from the current collector through a device being powered (cell phone, computer, etc.) to the negative current collector. The separator blocks the flow of electrons inside the battery.
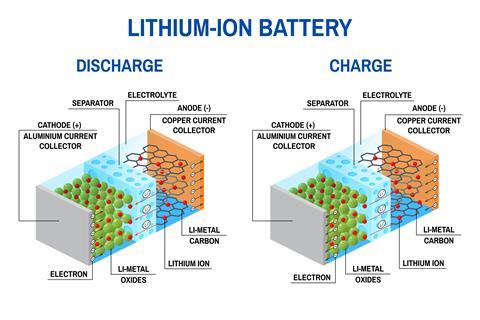
While the battery is discharging and providing an electric current, the anode releases lithium ions to the cathode, generating a flow of electrons from one side to the other. When plugging in the the truck to charge, the opposite happens: Lithium ions are released by the cathode and received by the anode.
The two most common concepts associated with batteries are energy density and power density. Energy density is measured in watt-hours per kilogram (Wh/kg) and is the amount of energy the battery can store with respect to its mass. Power density is measured in watts per kilogram (W/kg) and is the amount of power that can be generated by the battery with respect to its mass. You’ll hear a lot of people talking about both of these items in discussions about batteries and electric vehicles. I found this example from the people at the University of Calgary as a way of thinking about the difference:
”To better understand energy density, consider people lighting a fire while out camping. Naturally, the fire is first lit with kindling. It’s high surface area-to-volume ratio means that it burns quickly—a high power density. Once the fire is going, kindling isn’t a good fuel choice anymore, because it burns too fast. Now the fire burns better with logs because they have have a high energy density. A single log burns well for a long time.”
Having dabbled with Lithium metal rechargeable batteries during the 1980s, with disastrous effects - Lithium metal is very unstable and rechargeable batteries were prone to bursting into flames - the sector turned its attention to the Lithium-ion battery and Sony is generally credited as being the company which first commercialised the Lithium-ion battery. Today this chemistry has become the most promising and fastest growing battery on the market. Although lower in specific energy than lithium-metal, Lithium-ion is safe, provided the voltage and currents limits are respected.
Because of their high energy density, and their comparative lightness, stacking lots of lithium-ion cells together in the one place produces a battery pack far lighter and more compact than stacks made of other battery types. By stacking enough lithium-ion cells together it’s possible to reach sufficient voltage to power a battery electric HGV - hurrah, we have made it to the concept of the battery pack! With me so far…?
OK, so we understand how a battery works, but you’ll also probably be aware of people talking about different batter types - LFP, NMC for example. This is all referring to the make up of the cathode in the lithium-ion battery.
Before we move onto cathode chemistry, a minor detour to anode. It is usually graphite. However, the repeated insertion of lithium ions into the standard graphite structure in a typical lithium-ion battery eventually breaks apart the graphite. This reduces the battery’s performance and the graphite anode will eventually break down, and the battery will stop working. Researchers are working on options to use graphene (single-atom thick sheets of carbon) rather than graphite which will improve performance.
Anyway, back to the cathode as this appears to be where all the interest is currently. If things weren’t complicated enough with the shift to carbon zero vehicles, the type of cathode used in the battery is becoming a divergence point between the manufacturers. In our recent interview with Daimler Truck technical group manager Andreas Gorbach he explained how the German truck giant reckoned lithium iron phosphate batteries or LFPs were the best solution for trucks, others (Volta for example) are using NMCs. So, we thought we’d better give a quick heads up - thanks to the Battery University for giving the best overview we could find around the web.
The most common Lithium-ion battery types are Lithium Cobalt Oxide (LCO) typically used in mobile phones and laptops; Lithium Nickel Manganese Cobalt Oxide (NMC) which is the preferred choice for many passenger cars and some battery electric trucks and Lithium Iron Phosphate (LFP) which would appear to be increasing in popularity for the HGV sector. As you’d expect in this fast-moving period of development, opinions vary as to which is the best application for each battery particularly for NMC and LFP. NMC provides high capacity and high power, but the durability over the long life expected for trucks is questionable, while LFP is considered to have a longer life, but has lower energy density than NMC, so you’ll need more which will make the vehicle heavier.
And finally, we have solid state batteries - considered to be the nirvana of battery development. We’ve found a decent article from Ryoji Kanno, an Institute Professor at the Tokyo Institute of Technology if you want a bit more detail. Put simply, in a solid state battery the electrolyte (a liquid or polymer in a lithium-ion battery) is, as the name suggests, a solid. It is expected that the solid electrolyte will enable larger-capacity and higher-output batteries than lithium-ion batteries. By changing from a liquid to solid electrolyte, the ions will move better in the battery cell, making it possible to create batteries with larger capacity and higher output than lithium-ion batteries. Moreover, making the electrolyte solid has safety advantages over lithium-ion.
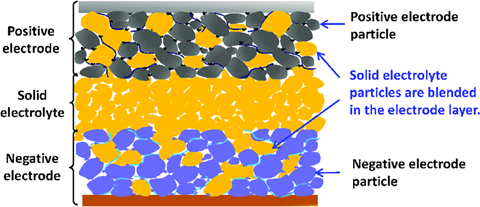
Offering fast-charging, high performance, long life and a high degree of flexibility in the shape of the battery, the automotive applications are obvious. But…there are still a number of challenges to overcome before solid state batteries can go into mass production and it could be more than 10 years before we see solid state batteries in heavy duty trucks. It will be worth the wait, though we can’t help wondering what next BIG thing will have emerged by then to confuse us even more.