Hydrogen fuel cell trucks are commercial vehicles that use a fuel cell to generate electricity to power their electric drivetrain. Like traditional internal combustion engine (ICE) trucks, they can be used for a variety of tasks, however, unlike ICE trucks, fuel cell trucks produce zero emissions at the tailpipe and are much quieter in operation.
The fuel cell itself is typically located on the vehicle’s chassis, where it converts hydrogen gas into electricity. The electricity powers an electric motor, which turns the wheels. The only bi-product of the process is water vapor.
Fuel cell trucks have the potential to reduce emissions and improve air quality in cities, as well as reduce dependence on fossil fuels. However, they are still relatively expensive to build and maintain, and the infrastructure for hydrogen fuel is not yet as widespread as for diesel.

The components of a Fuel Cell Electric Vehicle (FCEV) drivetrain
A fuel cell drivetrain is a powertrain system that uses a fuel cell to generate electricity to power an electric motor, which propels the vehicle.
The fuel cell drivetrain consists of several key components:
-
Fuel cell: This is the heart of the drivetrain, and it converts hydrogen gas into electricity through a chemical reaction. The fuel cell typically includes an anode, cathode, and electrolyte, which work together to create a current.
-
Hydrogen storage: The hydrogen gas used by the fuel cell must be stored in high-pressure tanks on the vehicle. These tanks are designed to withstand the pressure and keep the hydrogen gas safe and secure.
-
Electric motor: The electric motor is powered by the electricity generated by the fuel cell. It is connected to the vehicle’s wheels.
-
Battery: A small battery is also included in the drivetrain to store energy generated by the fuel cell, and to provide temporary power during high-demand situations such as acceleration.
-
Control system: A control system is used to manage the flow of hydrogen gas and electricity between the various components, and to ensure the fuel cell and electric motor are working together efficiently.
Fuel Cell technical explainer
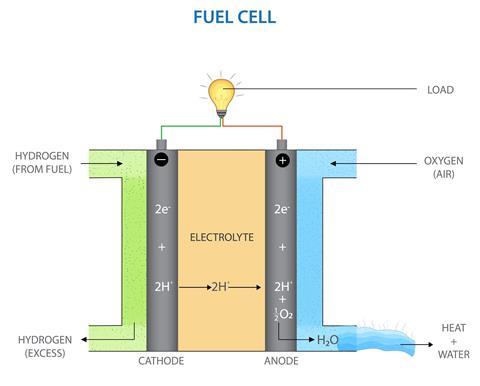
In a fuel cell, the anode and cathode are the two electrodes where the chemical reactions take place. The anode is the negative electrode and the cathode is the positive electrode.
The anode is where the fuel (such as hydrogen) enters the cell and is ionized, meaning the atoms are split into smaller particles. This process creates protons, which are positively charged, and electrons, which are negatively charged.
The protons are able to pass through the electrolyte, which is a material that allows only positively charged particles to pass through, and reach the cathode. The electrons, however, are forced to flow through an external circuit, creating a current. This is how electricity is produced in a fuel cell.
At the cathode, the protons and electrons are reunited with oxygen (from air) to form water, which is the only by-product of the reaction. The cathode is the positive electrode and is the site of the chemical reaction that consumes the hydrogen ions and electrons to produce water and heat.
Hydrogen can be produced in different ways
There are two main types of hydrogen that can be used in fuel cell electric vehicles (FCEVs): green hydrogen and grey hydrogen.
Green hydrogen is hydrogen that is produced through the process of electrolysis, using renewable energy sources such as solar or wind power to split water molecules into hydrogen and oxygen. This process is considered to be carbon-neutral, as the only bi-product is oxygen. Green hydrogen is considered as a sustainable and clean energy source.
Grey hydrogen, on the other hand, is hydrogen that is produced through steam methane reformation, which is a process that uses natural gas to produce hydrogen. This process releases carbon dioxide and other greenhouse gases, making it a carbon-emitting process. Grey hydrogen is the most common type of hydrogen produced today.
Brown hydrogen is another type of hydrogen is produced from coal. It is not considered as clean as the other two types.
It’s worth noting that hydrogen can also be produced from biogas, which is produced from organic waste. This type of hydrogen is considered to be sustainable and clean, as it is produced from a renewable source and can reduce the amount of waste that would otherwise be sent to landfill.
Fuel Cell Vehicles have a myriad of pros and cons
Fuel cell electric vehicles (FCEVs) have several advantages and disadvantages when compared to traditional internal combustion engine (ICE) vehicles and battery electric vehicles (EVs).
Pros of FCEVs:
- Longer driving range than battery EVs, which makes them suitable for long-distance travel and commercial use.
- Faster refuelling times than charging an EV, which can be an advantage for fleet operators and long-distance drivers.
- Zero tailpipe emissions, making them environmentally friendly and beneficial for air quality in cities.
- Hydrogen fuel can be produced from a variety of sources, including renewable energy, which means it has the potential to be carbon-neutral.
Cons of FCEVs:
- Currently, the infrastructure for hydrogen fuel is not as widespread as for diesel or electric charging.
- They are currently more expensive to build and maintain than ICE vehicles or battery EVs.
- Hydrogen fuel can be expensive, which can make FCEVs more costly to operate than other types of vehicles.
- The production and transportation of hydrogen can produce emissions if it is not produced using renewable energy sources.
- The storage of hydrogen fuel is a challenge, as it requires high-pressure tanks to prevent leakage or explosion.
Fuel cells have the potential to play an important role in the future of transportation and energy. The technology is still relatively new and has not yet been widely adopted, but it is seen as a promising alternative to traditional internal combustion engines and battery-electric powertrains.










